IEA Chief: "no silver bullet to cut greenhouse gas emissions"
 Speaking at a meeting of the UN's Economic Commission for Europe (UNECE) in which energy security, efficiency and climate change took central stage, International Energy Agency (IAE) chief Claude Mandil warned there was no "silver bullet" which by itself would cut greenhouse-gas emissions that drive dangerous climate change.
Speaking at a meeting of the UN's Economic Commission for Europe (UNECE) in which energy security, efficiency and climate change took central stage, International Energy Agency (IAE) chief Claude Mandil warned there was no "silver bullet" which by itself would cut greenhouse-gas emissions that drive dangerous climate change.Mandil set a target of an early cut of a billion tonnes of emissions per year and said a full range of measures - which he said included renewable energy, carbon storage, nuclear power and energy efficiency - should be harnessed. His statements come on the eve of the publication of the report on mitigating climate change to be released early May by Working Group III of the Intergovernmental Panel on Climate Change (IPCC).
The unsustainable present
Current fossil-fuel dominated energy demand increases carbon dioxide (CO2) by one billion tonnes every two years, according to IEA data. Energy demand will grow by more than 50 percent by 2030 if the pattern of consumption remains unchanged. "This is not sustainable," Mandil reiterated.
Even very basic energy savings like phasing out wasteful incandescent light bulbs from 2008 and better street lighting could make significant inroads, the IEA chief argued. The global cost of lighting could be reduced by 2.6 trillion dollars by 2030 and a cumulative 16 billion tonnes of carbon dioxide could be saved, according to the agency. "The difference could reach one-third of lighting costs in 2030," Mandil said. "The additional investment costs would easily be offset by consumption savings."
However, none of the technological steps could be implemented on the kind of scale needed to tackle greenhouse gas emissions on their own, he cautioned. About 78 percent of CO2 savings were likely to come from more efficient use of energy and 22 percent from cleaner energy sources by 2030, according to the agency.
Mandil said that to avoid one billion tonnes of greenhouse gases a year, the world would need to replace 300 convention coal-fired power plants with zero emission electricity generation every year, or build 150 one-gigawatt nuclear power plants. That is also the equivalent of multiplying the United States's current solar power capacity by about 1,300 every year or 200 times the US wind farm capacity.
Carbon capture and storage
So-called carbon capture and storage (CCS) techniques - which involve pumping carbon dioxide underground - are still largely untested and very costly with current technology. CCS is also contested in some scientific quarters, with experts fearing that storage chambers could be breached by earthquake or porous geology, spewing the CO2 into the atmosphere. (Which is why the Biopact thinks using CCS with carbon neutral biofuels is the safest way forward to test the technology - see our text at EurActiv; such a 'Bio-Energy with Carbon Storage' concept would also result in a carbon-negative energy system).
Mandil said 1,000 large carbon sequestration plants would have to be built annually to meet climate change targets. "But we're afraid that in 2030, carbon capture and sequestration technologies would not be available at an affordable cost on a large scale at that time," he said.
Thierry Desmarest, chairman of French oil giant Total which has a pilot CCS unit in Lacq in southwest France (earlier post), said it costs $100 a metric ton to capture and store CO2. An important objective would be to divide this cost by one-half or one-third, Desmarest told the forum in Geneva. CCS used in combination with biogas would be considerably less costly (previous post).
Biofuels to play major role
IEA chief Mandil said that biofuels had the potential to play a major role, but here again the challenge would be to reduce cost. "It will not be possible in the long term to sustain in the future with sustained subsidies," he said. Both Mandil and the IEA's Chief Economist earlier called on the EU and the US stop subsidizing their inefficient biofuels, to lift their trade barriers on imported biofuels, and to source the green fuels there where they can be produced cost-effectively and efficiently (earlier post):
 biofuels :: energy :: sustainability :: climate change :: greenhouse gases :: fossil fuels :: renewables ::ethanol :: biodiesel :: biomass :: bioenergy :: carbon negative :: carbon capture and storage :: IEA ::
biofuels :: energy :: sustainability :: climate change :: greenhouse gases :: fossil fuels :: renewables ::ethanol :: biodiesel :: biomass :: bioenergy :: carbon negative :: carbon capture and storage :: IEA :: Brazil and the United States, the two largest producers of ethanol, accounting for about 70 percent of world output, last month signed a broad agreement to work together to advance biofuels technology and set common standards for ethanol trade (earlier post).
Gregory Manuel, special advisor to Secretary of State Condoleezza Rice, said the United States was working with India, China, South Africa and the European Commission "to bring a standard to biofuels use such that the exponential potential for biofuels can be carried through with maximum efficiency". We reported on this recently launched effort, here.
The United States was looking at bringing the benefits of biofuels to certain countries, starting in the Western Hemisphere, but also in Africa and south-east Asia, he said. "The sustainability issues are critical and are integrated into that approach," Manuel said. The United States was also working with Brazil on research and development, "bringing second generation biofuels to the marketplace, hopefully faster than predicted," he said.
Global effort and US commitment needed
The chairman of French oil group Total, also addressing the meeting, welcomed Europe's role in spearheading movement on climate change but underlined the need for a global response. "That has to be welcomed, but that pioneering venture will only work if the United States and major emerging nations, which produce the most (greenhouse gas) emissions commit themselves to an equivalent effort," Thierry Demarest said.
The Total chief warned that unless that happened, the European effort could harm its industry's competitiveness in the longer term. The UN's Kyoto protocol set targets for industrialised countries to trim outputs of carbon dioxide (CO2) and other gases that trap solar heat, unbalancing the planet's delicate climate system, by 2012.
However, the United States and Australia have stayed out of the binding agreement, while efforts to draw up a post-2012 deal drawing in developing countries are mired in problems.
More information:
Reuters: No silver bullet to cut emissions: IEA chief - April 28, 2007.
France24: AFP News Brief: No silver bullet to combat climate change: IEA chief - April 28, 2007.
United Nations Economic Commission for Europe: Sixty-second session 25-27 April 2007, Palais des Nations, Geneva.
United Nations Economic Commission for Europe: Sustainable Energy Policies: the Key to Energy Security in the UNECE Region - Speakers Briefing Note [*.pdf], April 27, 2007.
Article continues
 --------------
--------------
 Taiwan's Feng Chia University has succeeded in boosting the production of hydrogen from biomass to 15 liters per hour, one of the world's highest biohydrogen production rates, a researcher at the university said Friday. The research team managed to produce hydrogen and carbon dioxide (which can be captured and stored) from the fermentation of different strains of anaerobes in a sugar cane-based liquefied mixture. The highest yield was obtained by the Clostridium bacterium.
Taiwan's Feng Chia University has succeeded in boosting the production of hydrogen from biomass to 15 liters per hour, one of the world's highest biohydrogen production rates, a researcher at the university said Friday. The research team managed to produce hydrogen and carbon dioxide (which can be captured and stored) from the fermentation of different strains of anaerobes in a sugar cane-based liquefied mixture. The highest yield was obtained by the Clostridium bacterium.



 Environmentally conscious shoppers may soon be able to cross non-recyclable packaging off their list as European researchers work towards reducing the carbon emissions caused by our throwaway society - with modified natural fibre based, intelligent packaging. Some of the plastics and materials they are developing combine rather exotic materials: a molecule found in the exoskeletons of crabs and crustaceans, clay-particles engineered on a nano-scale, DNA footprints to identify materials or photosensitive nano-particles that form organic screens to be used for communication capabilities.
Environmentally conscious shoppers may soon be able to cross non-recyclable packaging off their list as European researchers work towards reducing the carbon emissions caused by our throwaway society - with modified natural fibre based, intelligent packaging. Some of the plastics and materials they are developing combine rather exotic materials: a molecule found in the exoskeletons of crabs and crustaceans, clay-particles engineered on a nano-scale, DNA footprints to identify materials or photosensitive nano-particles that form organic screens to be used for communication capabilities.
 The ancient technique of burying charcoal into agricultural soils has gained attention over the years as a way to sequester carbon dioxide and fight climate change. Earlier we referred to scientists who are studying this tradition as it existed in the Amazon rainforest, where human-made and very fertile soils were discovered filled with char ("terra preta", "dark earth" -
The ancient technique of burying charcoal into agricultural soils has gained attention over the years as a way to sequester carbon dioxide and fight climate change. Earlier we referred to scientists who are studying this tradition as it existed in the Amazon rainforest, where human-made and very fertile soils were discovered filled with char ("terra preta", "dark earth" - 

 Biofuels are not destined for human consumption. For this reason, they can be grown on land not suitable for food crops. Earlier we referred to the potential of energy plantations to function as bioremediation agents on former industrial sites (previous post on
Biofuels are not destined for human consumption. For this reason, they can be grown on land not suitable for food crops. Earlier we referred to the potential of energy plantations to function as bioremediation agents on former industrial sites (previous post on  Nano Chemical Systems Holdings Inc.,
Nano Chemical Systems Holdings Inc.,  On 10 January 2007 the European Commission made proposals for a new Energy Policy for Europe (
On 10 January 2007 the European Commission made proposals for a new Energy Policy for Europe (
 Meeting in Berlin with former UN Secretary-General Kofi Annan and the UK's PM Tony Blair (who launched the Africa initiative at Gleneagles), German Chancellor Angela Merkel, who chairs both the G8 as well as the Presidency of the EU, said that the world's industrialized countries do not need to set new goals to help the developing world but keep the promises they have already made.
Meeting in Berlin with former UN Secretary-General Kofi Annan and the UK's PM Tony Blair (who launched the Africa initiative at Gleneagles), German Chancellor Angela Merkel, who chairs both the G8 as well as the Presidency of the EU, said that the world's industrialized countries do not need to set new goals to help the developing world but keep the promises they have already made.
 Biofuels are clearly 'value-free': both neoliberal free trade afficionados and anti-globalist organisations are investing heavily in the sector, as are moderate left-wing governments like those of Brazil and Chile as well as radical neo-communist and marxist states like Cuba and China or Burma and Venezuela. Joining the latter group is the
Biofuels are clearly 'value-free': both neoliberal free trade afficionados and anti-globalist organisations are investing heavily in the sector, as are moderate left-wing governments like those of Brazil and Chile as well as radical neo-communist and marxist states like Cuba and China or Burma and Venezuela. Joining the latter group is the  According to the
According to the 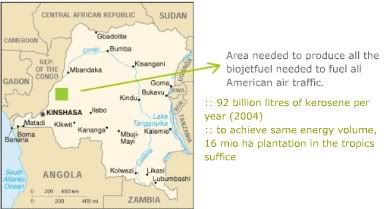

 Interesting news for the Biopact: the world's top international agriculture experts who met for three days at the UN's Food and Agriculture Organisation (FAO) headquarters in Rome to consider the environmental and food security impact of the rapidly-expanding bioenergy industry (
Interesting news for the Biopact: the world's top international agriculture experts who met for three days at the UN's Food and Agriculture Organisation (FAO) headquarters in Rome to consider the environmental and food security impact of the rapidly-expanding bioenergy industry (
 The
The 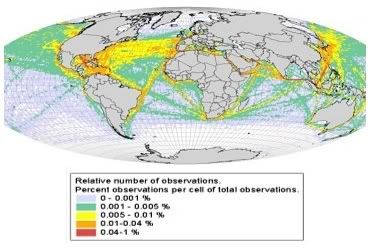
 Former EU Agriculture Commissioner Franz Fischler is less than optimistic about opportunities for the biofuel industry in Europe. Global competition from the South will prove to be too strong. The tropics and subtropics have much more available non-forest land, a good climate and highly productive crops such as sugarcane, palm oil, cassava and sorghum for liquid biofuels and eucalyptus, acacia and grass species for the production of solid biofuels. Green fuels made from those crops are competitive with oil and coal, whereas European biofuels need large amounts of subsidies to survive. Moreover, the EU does not have enough land, Fischler adds.
Former EU Agriculture Commissioner Franz Fischler is less than optimistic about opportunities for the biofuel industry in Europe. Global competition from the South will prove to be too strong. The tropics and subtropics have much more available non-forest land, a good climate and highly productive crops such as sugarcane, palm oil, cassava and sorghum for liquid biofuels and eucalyptus, acacia and grass species for the production of solid biofuels. Green fuels made from those crops are competitive with oil and coal, whereas European biofuels need large amounts of subsidies to survive. Moreover, the EU does not have enough land, Fischler adds.
 The energy balance and the greenhouse gas (GHG) balance of biofuels are two important criteria to judge whether these fuels are energetically interesting and whether they can be a weapon in the fight against climate change. We already know that fuels made from crops grown in the South have both a very strong energy balance as well as a GHG balance that proves they substantially reduce emissions of most climate destructive gases. For biofuels made from crops like rapeseed or corn, the situation looks more problematic.
The energy balance and the greenhouse gas (GHG) balance of biofuels are two important criteria to judge whether these fuels are energetically interesting and whether they can be a weapon in the fight against climate change. We already know that fuels made from crops grown in the South have both a very strong energy balance as well as a GHG balance that proves they substantially reduce emissions of most climate destructive gases. For biofuels made from crops like rapeseed or corn, the situation looks more problematic. The UN's
The UN's 








Saturday, April 28, 2007
New class of enzymes discovered that could increase efficiency of cellulosic ethanol production
Biofuels such as corn ethanol or rapeseed biodiesel are unsustainable, because they have a very weak energy balance and do not reduce greenhouse gas emissions much. For this reason, scientists are looking into developing 'second-generation' biofuels, made from cellulose, which is a far more abundant feedstock than the oil and sugar currently obtained from grains, canes and oil seeds. Cellulose is the most abundant biological material on earth.
Even though 'first-generation' biofuels made from tropical feedstocks, such as sugarcane or cassava already do have a strong energy balance and reduce CO2 emissions considerably, utilizing the cellulosic waste biomass obtained during their production as a feedstock for ethanol would likewise increase these balances even further and make tropical biofuels extremely energy efficient.
However, the production of biofuels from cellulose in mass quantities is still quite costly. The development of thermochemical conversion technologies - which involve gasification or pyrolysis of biomass - is making good progress, even though cost and downstream processing into useable fuels remains an obstacle. The same is true for the biochemical conversion path, which is based on the use of enzymes to break down the cell walls of plants and to release the sugars they contain that can then be fermented into alcohol.
With current technologies, this critical step of breaking down cell walls relies on microbial enzymes called "cellulases" to digest the cellulose. The microbial enzymes have a structure that makes them very efficient at binding to and digesting plant cell wall material called lignocellulose (a combination of lignin and cellulose).
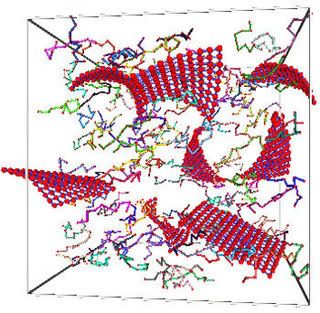
But now, a new class of plant enzymes with a similar structure has been discovered, potentially offering researchers new properties for producing ethanol even more efficiently. A team working with Jocelyn Rose, Cornell assistant professor of plant biology, published its paper [*abstract] on the new class of enzymes in the April 20 issue of the Journal of Biological Chemistry. Breeanna Urbanowicz, a graduate student in Rose's laboratory, was the paper's lead author.
For an enzyme to break down cellulose, a structure called a cellulose-binding module attaches to the cellulose. Once attached, a catalyst then breaks the cell wall material into small units, which can then be turned into ethanol. While researchers have known that plants have cellulase-like enzymes, it was previously thought that they did not have a cellulose-binding module, and so could not attach to cellulose or digest it very effectively -- until now:
"This is the first example of a cellulose-binding domain in a plant cell wall enzyme," said Rose. While the new enzyme was found in a tomato plant, Rose and colleagues have evidence of a set of such plant proteins in many species that potentially could be used for biofuel production. Biofuel research may also help uncover exciting new uses for these enzymes, said Rose. Researchers may, for example, breed for plants with high levels of these proteins.
Though the scientists stress that more study is needed to understand how plants use this class of enzymes, Rose speculates that they may be needed when growing tissues rapidly expand and require loosening of tightly bound strands of cellulose, called microfibrils, that make up a cell wall's structure. The binding enzymes may also be part of the process of breaking down tissues, e.g., when fruits -- such as tomatoes -- soften.
Among others, co-authors included Carmen Catalá, a research associate previously working in the Department of Plant Biology, who originally identified the gene for the tomato enzyme, and David Wilson, Cornell professor of molecular biology and genetics.
Image: This schematic diagram shows the newly discovered class of plant enzymes with a cellulose-binding module (shown in blue), sticking to a plant cell wall. The binding module of the enzyme helps the catalytic region of the enzyme (shown in more detail in gray in the pullout part of the picture) break down the crystalline cellulose. Courtesy: Daniel Ripoll and Chris Pelkie/Cornell Theory Center.
More information:
Breeanna R. Urbanowicz, Carmen Catalá, Diana Irwin, David B. Wilson, Daniel R. Ripoll, and Jocelyn K. C. Rose, "A Tomato Endo-beta-1,4-glucanase, SlCel9C1, Represents a Distinct Subclass with a New Family of Carbohydrate Binding Modules (CBM49)" [*abstract], J. Biol. Chem., Vol. 282, Issue 16, 12066-12074, April 20, 2007
Cornell University: Newly discovered plant enzymes could lead to more efficient -- and less costly -- ethanol production from cellulose - April 24, 2007.
Article continues
posted by Biopact team at 9:49 PM 0 comments links to this post