Rationale behind the International Biofuels Forum - a new energy paradigm
 The International Biofuels Forum, created by Brazil, the European Union, the United States, India and China to promote the use of biofuels, has officially been installed. Senior diplomats from participating countries gathered to launch the project at the United Nations which said that the forum will contribute to creating a world market for alternative fuels, resulting in economic, social and environmental benefits for developed and developing countries alike.
The International Biofuels Forum, created by Brazil, the European Union, the United States, India and China to promote the use of biofuels, has officially been installed. Senior diplomats from participating countries gathered to launch the project at the United Nations which said that the forum will contribute to creating a world market for alternative fuels, resulting in economic, social and environmental benefits for developed and developing countries alike.The forum will initially be established for one year and meet regularly to discuss ways to promote the sustained use and production of biofuels around the globe.
An overview of comments that capture the rationale behind the initiative:
On energy security and global development
Biofuels constitute a viable economic alternative for the partial substitution of fossil fuels and the diversification of the world’s energy mix. This initiative creates a mechanism to structure the dialogue among the biggest producers and consumers of biofuels.
The introduction of biofuels would benefit developed countries through increasing energy security by reducing the dependence on fossil fuels and contributing to lower greenhouse gas emissions. For developing countries, greater use of biofuels would significantly reduce dependence on imported oil, redressing trade imbalances and freeing up income for investments in health, education and social programmes. -Antonio Patriota, Ambassador of Brazil to the United States.
On the new energy paradigm
Access to affordable energy is fundamental to economic and social development. We understand the international biofuels forum and the efforts of all of our partners in addressing the issue of biofuels to be not just an issue of energy security, not just an environmental issue, but fundamental to how we are going to address basic economic and social development problems, and recognizing that by working together we will be able to identify ways to help countries with agricultural productive potential to be become major energy suppliers.
This a huge step forward in the development of a new international understanding of energy.
-Thomas A. Shannon, Jr., United States Assistant Secretary of State for Western Hemisphere Affairs.
On climate change and greenhouse gases
The development and sustained use of biofuels will contribute to social and economic development and improve the world’s energy structure, diversifying energy sources. It’s also important for the international efforts aimed at mitigating climate change by reducing greenhouse gas emissions.
-Liu Zhenmin, deputy Permanent Representative of China to the United Nations
On job creation, social and rural development
Another objective of the biofuels initiative is job creation. We want to ensure that previously disadvantaged people and people in rural communities will be close to where the biofuels initiative will take place, and they will receive the benefits of this initiative.Asked whether the Forum would only be able to make recommendations or if it would have enforcement power, Mr. Patriota said he wished to clarify that the Forum was not a new international organization, but rather a mechanism for closer coordination among the field’s major players to establish common standards and work towards the commoditization of biofuels, so that they might eventually be traded like oil. The forum would continue to work in the six-party format for the time being, but, in the future, would bring in other players, he said:
- Elsa du Toit, Director, Energy Efficiency, Department of Minerals and Energy of South Africa.
 biomass :: bioenergy :: energy :: sustainability :: climate change :: rural development :: social development :: poverty alleviation :: trade :: environment :: ethanol :: biodiesel :: biofuels :: International Biofuels Forum :: United Nations ::
biomass :: bioenergy :: energy :: sustainability :: climate change :: rural development :: social development :: poverty alleviation :: trade :: environment :: ethanol :: biodiesel :: biofuels :: International Biofuels Forum :: United Nations :: In response to questions about why the Forum did not include representation from the Middle East or the Russian Federation, and whether those countries would eventually be included in negotiations, Mr. Patriota said that Brazil and the Russian Federation were currently discussing the matter bilaterally and that Middle Eastern countries would be represented as part of Asia or Africa.
“This is not a closed club,” he added. “We would welcome very much contacts with other nations across the globe.”
In response to a question about how much of the fuel consumed in the world might eventually come from biofuels, Eduardo Pereira de Carvalho, the President of UNICA, a group representing 70 per cent of ethanol producers in Brazil, said that 2 or 3 per cent was most likely initially. That figure could someday rise to as high as 30 per cent, however. Given his expectation that oil prices would climb in the coming years, he said that biofuels were an “important participant of the new energy matrix of the twenty-first century”.
“What we are doing is much more than responding to high gasoline or oil prices,” he added, noting that the Biofuels Forum was aimed at addressing fuel supply security, global warming and the use of biofuels as an instrument for development.
Brazil's competitiveness
The establishment of the Forum comes a week before President Bush travels to Brazil for talks in which renewable biofuels will top the agenda. Bush and Brazilian President Luiz Inacio Lula da Silva are expected to sign an agreement -- separate from the international forum -- to share scientific and technological research for an industry that has grown in both countries.
Brazil is the world's biggest ethanol exporter, using sugar cane to produce it. Eight out of 10 Brazilian cars run on ethanol, which emits far less greenhouse gases than fossil fuels. In the United States, where farmers use corn to make ethanol, production surpasses that in Brazil.
The promotion of ethanol could eventually help wean the U.S. off its need for foreign oil, officials say, lessening the energy dependence on volatile Middle Eastern nations and Venezuela -- whose President Hugo Chavez has long been a political thorn in the Bush administration's side.
But teaming up with Brazil on the promotion of ethanol hasn't pleased everyone: Corn farmers in the U.S. don't like the idea of the government helping Brazil's industry, which they see as a competitor. Lawmakers from corn-growing states have registered their complaints with Bush.
But Silva said the issue of moving the world off of fossil fuels was more important than any bickering over a Brazil-U.S. agreement.
"Biofuel cannot be promoted by just one country," Silva told journalists in Brazil on Thursday. "It is a global issue."
Brazil's method of producing ethanol is better than the American way, Silva suggested, noting that sugarcane-based ethanol is far cheaper to make than corn-based ethanol, and warm-weather climates like Brazil are the only places where sugar cane thrives.
But neither country produces enough ethanol to meet growing domestic demand. And while countries from Asia to Europe are pursuing biofuels as a way to reduce dependency on oil imports, international trade of alternative fuels is minimal.
A Brazilian industrial group estimates that current ethanol usage amounts to only two percent of world oil consumption. Brazil, with its vast land resources, is hoping to produce enough ethanol to satisfy 10 percent of the world's demand for gasoline within the next 20 years.
President Bush has set a goal for the United States to use 130 billion liters of alternative fuels a year by 2017. Current capacity is about 20 billion liters a year.
More information:
Biofuel Review: UN launches the International Biofuels Forum - March 2, 2007
Business Week: Forum to promote biofuels trade debuts - March 3, 2007
The Hindu: International forum to promote biofuel launched - March 3, 2007
Article continues
 --------------
--------------
 Taiwan's Feng Chia University has succeeded in boosting the production of hydrogen from biomass to 15 liters per hour, one of the world's highest biohydrogen production rates, a researcher at the university said Friday. The research team managed to produce hydrogen and carbon dioxide (which can be captured and stored) from the fermentation of different strains of anaerobes in a sugar cane-based liquefied mixture. The highest yield was obtained by the Clostridium bacterium.
Taiwan's Feng Chia University has succeeded in boosting the production of hydrogen from biomass to 15 liters per hour, one of the world's highest biohydrogen production rates, a researcher at the university said Friday. The research team managed to produce hydrogen and carbon dioxide (which can be captured and stored) from the fermentation of different strains of anaerobes in a sugar cane-based liquefied mixture. The highest yield was obtained by the Clostridium bacterium.


 Thermoset resins are some of the most commonly used materials in the construction, furniture and automotive industries. World demand currently stands at around 25 million tonnes per year for the product that is used to make a wide variety of composite materials from particleboard to glass fibre panels and fighter jet wings.
Thermoset resins are some of the most commonly used materials in the construction, furniture and automotive industries. World demand currently stands at around 25 million tonnes per year for the product that is used to make a wide variety of composite materials from particleboard to glass fibre panels and fighter jet wings. The European Commission's ambitious proposal to increase the EU's share of renewable energies to a mandatory 20% of the bloc's overall consumption by 2020 (
The European Commission's ambitious proposal to increase the EU's share of renewable energies to a mandatory 20% of the bloc's overall consumption by 2020 ( Sergio Marini, the newly appointed president of the Italian farming industry association
Sergio Marini, the newly appointed president of the Italian farming industry association  As part of a series of bilateral agreements on 'mutual development' towards the 'Bolivarian Alternative', Cuba and Venezuela have
As part of a series of bilateral agreements on 'mutual development' towards the 'Bolivarian Alternative', Cuba and Venezuela have  Wang Hongguang, Director-General of the
Wang Hongguang, Director-General of the  Secondly, "to increase farmers' incomes. To develop biomass energy can create employment opportunities and bring more incomes to farmers. It is briefly estimated that to create a "green Daqing Oilfield" is equivalent to give RMB 120~150 billion yuan [€11.8-14.7/US15.5-19.4 billion] originally used to import petroleum to farmers and biomass energy enterprises while 12~15 million jobs could be thus created."
Secondly, "to increase farmers' incomes. To develop biomass energy can create employment opportunities and bring more incomes to farmers. It is briefly estimated that to create a "green Daqing Oilfield" is equivalent to give RMB 120~150 billion yuan [€11.8-14.7/US15.5-19.4 billion] originally used to import petroleum to farmers and biomass energy enterprises while 12~15 million jobs could be thus created."
 The
The 
 On Friday, Brazil, the European Union, the United States, South Africa, China and India will formally establish the 'International Biofuels Forum'. The idea is to create common norms and standards for bioenergy products, especially ethanol, to consolidate and facilitate world trade in the rapidly growing sector. The initiative behind the new international organisation, the offices of which will be based at the United Nations (UN) in New York, was taken by Brazil, the world's largest biofuels producer.
On Friday, Brazil, the European Union, the United States, South Africa, China and India will formally establish the 'International Biofuels Forum'. The idea is to create common norms and standards for bioenergy products, especially ethanol, to consolidate and facilitate world trade in the rapidly growing sector. The initiative behind the new international organisation, the offices of which will be based at the United Nations (UN) in New York, was taken by Brazil, the world's largest biofuels producer. Earlier we reported that advances in biotech, materials sciences and biofuels may soon make it possible to manufacture cars entirely made out of bio-based products: durable bioplastic fuel lines, car seat foams, body panels, structural components, interior parts and tires have already been developed that consist almost exclusively of bio-degradable, renewable, plant-based materials and no longer of petroleum (
Earlier we reported that advances in biotech, materials sciences and biofuels may soon make it possible to manufacture cars entirely made out of bio-based products: durable bioplastic fuel lines, car seat foams, body panels, structural components, interior parts and tires have already been developed that consist almost exclusively of bio-degradable, renewable, plant-based materials and no longer of petroleum ( The U.S. Department of Energy (DOE)
The U.S. Department of Energy (DOE)  Atmospheric scientists have uncovered fresh evidence to support the hotly debated theory that global warming has contributed to the emergence of stronger hurricanes in the Atlantic Ocean.
Atmospheric scientists have uncovered fresh evidence to support the hotly debated theory that global warming has contributed to the emergence of stronger hurricanes in the Atlantic Ocean. What can now be called 'the Brazilian dilemma' is becoming ever sharper. New studies show that global warming may make a far larger number of Brazil's enormously diverse plant and animal species go extinct, than previously thought. At the same time, Brazil is the leader in the production of climate-neutral biofuels the use of which does precisely what is needed: to reduce the carbon dioxide emissions responsible for global warming. However, large-scale biofuel production itself affects the biodiversity of ecosystems, because it is mostly based on monocultures.
What can now be called 'the Brazilian dilemma' is becoming ever sharper. New studies show that global warming may make a far larger number of Brazil's enormously diverse plant and animal species go extinct, than previously thought. At the same time, Brazil is the leader in the production of climate-neutral biofuels the use of which does precisely what is needed: to reduce the carbon dioxide emissions responsible for global warming. However, large-scale biofuel production itself affects the biodiversity of ecosystems, because it is mostly based on monocultures.
 One of the world's top climate scientists, geoscientist James Hansen delivered an address to the National Press Club, in Washington, D.C., calling for the United States to end the building of new coal-fueled power plants and begin dismantling those in operation now that do not scrub high levels of carbon dioxide emissions. Hansen is the director of
One of the world's top climate scientists, geoscientist James Hansen delivered an address to the National Press Club, in Washington, D.C., calling for the United States to end the building of new coal-fueled power plants and begin dismantling those in operation now that do not scrub high levels of carbon dioxide emissions. Hansen is the director of  State news agency
State news agency  NanoDynamics, Inc., a diversified nanotechnology and manufacturing company,
NanoDynamics, Inc., a diversified nanotechnology and manufacturing company,  In the context of the Kyoto Protocol, the EU has committed to reducing greenhouse-gas emissions by 8%, compared with 1990 levels, before 2012.
In the context of the Kyoto Protocol, the EU has committed to reducing greenhouse-gas emissions by 8%, compared with 1990 levels, before 2012.  Tonight, Europe's leading cultural television channel and production house, ARTE, airs a documentary on biofuels in Brazil (presentation of the film in
Tonight, Europe's leading cultural television channel and production house, ARTE, airs a documentary on biofuels in Brazil (presentation of the film in  But even in the case of a green, renewable, and sustainably produced energy source, the picture is not all that rosy. Until recently, the main beneficiaries of the industry have been the large agro-industrial firms who enjoy scale-advantages and rely on highly mechanised production. However, the situation is changing gradually in favor of smallholders. Brazil's landless people, united in Latin America's largest social movement, the 'Movimento dos Trabalhadores Rurais Sem Terra', are beginning to enjoy the benefits of the industry's expansion, because they have found a listening ear in Brazil's recently re-elected president Luis Inácio Lula da Silva.
But even in the case of a green, renewable, and sustainably produced energy source, the picture is not all that rosy. Until recently, the main beneficiaries of the industry have been the large agro-industrial firms who enjoy scale-advantages and rely on highly mechanised production. However, the situation is changing gradually in favor of smallholders. Brazil's landless people, united in Latin America's largest social movement, the 'Movimento dos Trabalhadores Rurais Sem Terra', are beginning to enjoy the benefits of the industry's expansion, because they have found a listening ear in Brazil's recently re-elected president Luis Inácio Lula da Silva. The African oil palm is a crop that yields an enormous amount of biomass (
The African oil palm is a crop that yields an enormous amount of biomass ( In a development of major importance for the future of the biobased economy, plant biologists at the University of Connecticut have
In a development of major importance for the future of the biobased economy, plant biologists at the University of Connecticut have 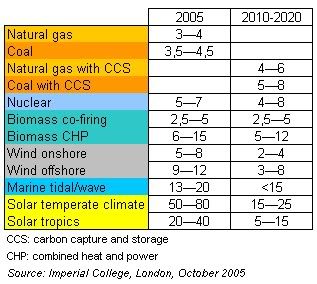 Some notes have to be added to this projection. First of all, the data are from a
Some notes have to be added to this projection. First of all, the data are from a  Two leading car manufacturers will be present at the
Two leading car manufacturers will be present at the  Senegal is the only country in West-Africa not to have suffered civil war or coup d'état since it became independent from France. Today, the Senegalese elect their new leader in a well organised democratic election. The incumbent, president Abdoulaye Wade, is widely expected to win in the first round, against 14 competitors.
Senegal is the only country in West-Africa not to have suffered civil war or coup d'état since it became independent from France. Today, the Senegalese elect their new leader in a well organised democratic election. The incumbent, president Abdoulaye Wade, is widely expected to win in the first round, against 14 competitors.






Saturday, March 03, 2007
Genome sequencing reveals how bacterium selects enzymes for cellulase, key to viable ethanol production
Cellulose-rich waste products from agriculture and forestry, such as grass clippings and wood chips — once thought too difficult to turn into ethanol — may soon be fodder for hungry, gene-tweaked bacteria who convert the biomass into useful bio-products and fuels.
The findings in today's Proceedings of the National Academy of Sciences may empower scientists to engineer ethanol-producing 'super-organisms' that can make clean-burning fuel from the billions of tons of biomass that are produced each year and that remain unused.
Ethanol holds the promise of a clean, renewable alternative to fossil fuels, but deriving it from ligno-cellulose is difficult. Producing it from corn is the easiest method, but doing so on a large scale would drive up the price of corn, corn starch, and even tangential foods like beef, since cows are fed on corn—not to mention all the energy spent fertilizing, maintaining, and harvesting a crop like corn. Conversely, deriving ethanol from plant materials such as the corn stalks and wood chips is challenging because the plants' cellulose is a very tough substance to break down, making for an inefficient process:
Wu's technique may prove much more effective than traditional methods. Instead of using separate steps to break down biomass into glucose and ferment the glucose into ethanol, as is currently done, Wu is working on a way to make a bacterium break down and ferment plant biomass efficiently in just one step.
Wu investigated C. thermocellum, which is a microorganism that has that ability to turn biomass into ethanol in one step, but is not used at the industrial scale yet because the first step, breaking down the plant's cellulose, is much too inefficient. The key, Wu surmised, is to find out what enzymes the bacterium uses to accomplish its feat, and then boost its ability to produce those enzymes. The problem, however, lies in the fact that C. thermocellum uses more than 100 enzymes, and any of the millions of combinations of them may be the magic mixture to break down a particular biomass.
So, Wu decided to make the bacterium do the work for him.
"The bacteria know how to express just the right genes to break down any particular biomass substrate, and we wanted to know how they know to turn on and off just the right genes at the right time to do the trick," says Wu. "We found the bacterium essentially throws the whole bowl of spaghetti at the wall, sees what sticks, and then makes a lot of that particular noodle."
C. thermocelllum produces low levels of many of its enzymes at any one time. When the bacterium comes in contact with wood, for instance, a few of its enzymes break down some of that wood. A product of that tiny reaction is a sugar called laminaribiose that diffuses into the cell. There it deactivates a repressor for two genes, which wake up and start pumping out the two triggers the full production of wood-degrading enzymes CelC and LicA.
Wu's paper shows the first time the triggering pathway for enzyme production in this bacterium has been revealed, and it was only possible because C. thermocellum genome was just recently sequenced, thanks to Wu's collaboration with the U. S. Department of Energy. With its 100 busy enzymes, the entire genome had to be observed as a whole, since fiddling with combinations of two, three, or more enzymes at a time would have taken "more than our lifetime," Wu says.
Wu is now working to re-engineer C. thermocellum to express an abundance of particular genes so it can readily and efficiently produce ethanol from a particular biomass. He's also continuing the genome-wide search for enzyme combinations that will degrade and ferment grasses, corn stovers, and even food waste.
"I don't think this is the revolution that makes ethanol a mainstay," says Wu, "but I believe this is a part of what will lead to the revolution."
Image: The bacterium Clostridium thermocellum.
More information:
Michael Newcomb, Chun-Yu Chen, and J. H. David Wu, Induction of the celC operon of Clostridium thermocellum by laminaribiose, [*abstract], February 27, 2007 Proc. Natl. Acad. Sci. USA, 10.1073/pnas.0700087104.
Article continues
posted by Biopact team at 2:35 PM 0 comments links to this post