Ocean acidification worse than expected, threatens sea life
Ocean acidification worse than expected, threatens sea life
mongabay.com
May 22, 2008
|
|
Increasing ocean acidification along the continental shelf of North America will likely have negative impacts on marine ecosystems, including the corrosion of calcium carbonate exoskeletons in many organisms, warn researchers writing in the journal Science.
Surveying waters from central Canada to northern Mexico, Richard A. Feely of the National Oceanic and Atmospheric Administration’s (NOAA) Pacific Marine Environmental Laboratory and colleagues found lower pH levels in seawater closer to the surface than expected. The authors attribute the acidification to the ocean’s increased absorption of carbon dioxide from the atmosphere. Anthropogenic emissions of CO2 are driving the trend.
“Since the beginning of the industrial era, the oceans have absorbed approximately 127 ± 18 billion metric tons of carbon as carbon dioxide from the atmosphere, or about one third of the anthropogenic carbon emissions released,” the authors write. “This process of absorption of anthropogenic CO2 has benefited humankind by significantly reducing the greenhouse gas levels in the atmosphere and minimizing some of the impacts of global warming. However, the ocean’s daily uptake of 30 million metric tons of carbon dioxide is significantly impacting its chemistry and biology.”
“Our results show for the first time that a large section of the North American continental shelf is impacted by ocean acidification.”
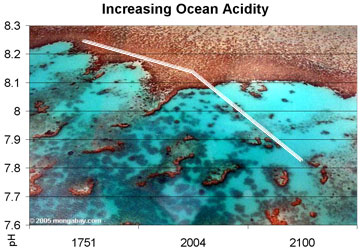 Observed and projected decline in global ocean pH, 1750-2100. Photo by Rhett A. Butler |
Since the industrial revolution, ocean pH levels have fallen by about 0.1 units but estimates based on Intergovernmental Panel on Climate Change (IPCC) emission scenarios suggest that surface water pH levels could drop by 0.4 pH units. The rapid decline would have dramatic impacts on sea life by reducing the availability of carbonate ions necessary for the formation of shells and exoskeletons. Corals, marine plankton, and shellfish would be especially affected, although some research has suggested that even juvenile fish would suffer from higher-than-normal CO2 levels. The authors say more studies are needed to determine the impact of ocean acidification on a wider range of species and ecosystems, but suggest that the ecological changes wrought by acidic conditions may have significant economic consequences, especially for marine fisheries. Many commercial fish species rely on plankton and other carbonate-dependent organisms for sustenance.
Because ocean acidification is largely independent of temperature, some scientists have warned that schemes to “geoengineer” Earth’s climate by blocking sunlight or increasing the reflectivity of the plant’s surface would not solve the problem. Further, since there is a lag time in the absorption of carbon dioxide by oceans, the impact of past carbon emissions are not yet fully apparent
R.A. Feely, C.L. Sabine, J.M. Hernandez-Ayon, D. Ianson, B. Hales (2008). “Evidence for Upwelling of Corrosive ‘Acidified’ Water onto the Continental Shelf,” Scienc 22 May 2008.
More articles on Ocean Acidification