Nitrogen pollution harming ecosystems and contributing to global warming
Nitrogen pollution harming ecosystems and contributing to global warming
mongabay.com
May 15, 2008
|
|
Nitrogen pollution of the world’s oceans is harming marine ecosystems and contributing to global warming, report two reviews published in the journal Science.
The research, which involved dozens of scientists from around the world, shows that human activity is dramatically altering nitrogen cycles in Earth’s oceans, soils, and atmosphere. The papers report that agricultural runoff and the burning of fossil fuels have boosted the supply of reactive nitrogen in the open oceans 50 percent above the normal range.
The first paper, led by Robert Duce of Texas A&M University, found that nitrogen produced by human activity is responsible for about a third of the nitrous oxide and a tenth of the carbon dioxide input to the world’s oceans each year. The researchers say the excess nitrogen “can deplete essential oxygen levels in the water and has significant effects on climate, food production, and ecosystems all over the world,” according to a statement from Science.
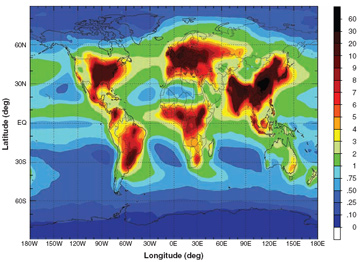 Estimated N deposition from global total N (NOy and NHx) emissions, totaling 105 Tg N per year. Courtesy of Science. |
Duce and colleagues calculate that humans account for up to three percent of the new marine biological production annually. While the increased biological activity sequesters CO2 from the atmosphere, the process produces nitrous oxide (N20), a greenhouse gas far more potent than carbon dioxide.
“This fertilization of the ocean by human activities has an important impact on the exchange of the greenhouse gases carbon dioxide and nitrous oxide and should be considered in future climate change scenarios,” Duce, a professor at Texas A&M University, said.
“Anyone concerned about climate change will be alarmed at the scale of man’s impact on the world’s oceans, as revealed by our new study,” added Peter Liss, a co-author of the paper and an environmental scientist at the University of East Anglia. “The natural nitrogen cycle has been very heavily influenced by human activity over the last century — perhaps even more so than the carbon cycle — and we expect the damaging effects to continue to grow. It is vital that policy makers take action now to arrest this.
“The solution lies in controlling the use of nitrogen fertilizer and tackling pollution from the rapidly increasing numbers of cars, particularly in the developing world,” he continued.
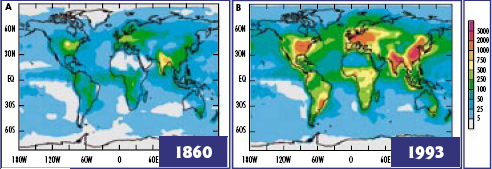 Nitrogen Deposition Past and Present mg N/m2/yr In 1860, the total Nr deposition to the Earth’s surface was 32 million metric tons of nitrogen, mostly from natural emission sources. By the early 1990s, total Nr deposition had increased to 100 million metric tons. The difference was entirely due to anthropogenic activities. In some regions, deposition increased 100-fold. Adapted from Galloway et al, 2004 and appearing at initrogen.org. Caption quoted from Human Alteration of the Nitrogen Cycle: Threats, Benefits, and Opportunities, a Joint UNESCO-SCOPE-INI Policy Brief. |
In the second paper, James N. Galloway of the University of Virginia and colleagues highlight health and environmental problems that arise from increased nitrogen pollution. The authors note an “extreme imbalance” of nitrogen and argue for the need to reduce the amount of reactive nitrogen in the environment.
“The public does not yet know much about nitrogen, but in many ways it is as big an issue as carbon, and due to the interactions of nitrogen and carbon, makes the challenge of providing food and energy to the world’s peoples without harming the global environment a tremendous challenge,” Galloway explained. “We are accumulating reactive nitrogen in the environment at alarming rates, and this may prove to be as serious as putting carbon dioxide in the atmosphere.”
“Nitrogen is needed to grow food but because of the inefficiencies of nitrogen uptake by plants and animals, only about 10 to 15 percent of reactive nitrogen ever enters a human mouth as food. The rest is lost to the environment and injected into the atmosphere by combustion,” he continued. “We must soon begin to manage nitrogen use in an integrated manner by decreasing our rate of creation of reactive nitrogen while continuing to produce enough food and energy to sustain a growing world population.”
CITATIONS:
J.N. Galloway et al (2008). “Transformation of the Nitrogen Cycle: Recent Trends, Questions, and Potential Solutions.” Science 16 May 2008
R.A. Duce t al (2008). “Impacts of Atmospheric Anthropogenic Nitrogen on the Open Ocean.” Science 16 May 2008