Scientists: aggressive CO2 reductions needed now to counter destruction of marine life
How much carbon dioxide is too much? According to United Nations Framework Convention on Climate Change (UNFCCC) greenhouse gases in the atmosphere need to be stabilized at levels low enough to "prevent dangerous anthropogenic interference with the climate system." But scientists have come to realize that an even more acute danger than climate change is lurking in the world's oceans — one that is likely to be triggered by CO2 levels that are modest by climate standards.
Ocean acidification could devastate coral reefs and other marine ecosystems even if atmospheric carbon dioxide stabilizes at 450 ppm, a level well below that of many climate change forecasts, report chemical oceanographers Long Cao and Ken Caldeira of the Carnegie Institution's Department of Global Ecology in the journal Geophysical Research Letters.
The researchers' conclusions are based on computer simulations of ocean chemistry stabilized at atmospheric CO2 levels ranging from 280 parts per million (pre-industrial levels) to 2000 ppm. Present levels are 380 ppm and rapidly rising due to accelerating emissions from human activities, primarily the burning of fossil fuels.
This study was initiated as a result of Caldeira's testimony [*.pdf] before a Congressional subcommittee on Fisheries, Wildlife and Oceans in April of 2007. At that time he was asked what stabilization level would be needed to preserve the marine environment, but had to answer that no such study had yet addressed that question. Cao and Caldeira's study helps fill the gap.
Atmospheric CO2 absorbed by the oceans' surface water produces carbonic acid, the same acid that gives soft drinks their fizz, making certain carbonate minerals dissolve more readily in seawater. This is especially true for aragonite, the mineral used by corals and many other marine organisms to grow their skeletons. For corals to be able to build reefs, which requires rapid growth and strong skeletons, the surrounding water needs to be highly supersaturated with aragonite.
 energy :: sustainability :: biomass :: bioenergy :: fossil fuels :: carbon dioxide :: ocean acidification :: marine biodiversity :: oceans ::
energy :: sustainability :: biomass :: bioenergy :: fossil fuels :: carbon dioxide :: ocean acidification :: marine biodiversity :: oceans ::
If current trends in CO2 emissions continue unabated, says Caldeira, in the next few decades, we will produce chemical conditions in the oceans that have not been seen for tens of millions of years. We are doing something very profound to our oceans. Ecosystems like coral reefs that have been around for many millions of years just won't be able to cope with the change.
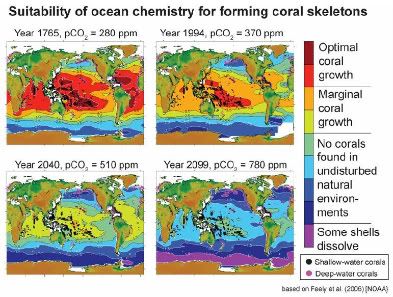
Figure: Maps showing the distribution of ocean chemistry suitable for coral growth for different time periods, assuming “business-as-usual” CO2 emissions. Colors represent the chemical force promoting the development of coral skeletons.
Year 1765: Several hundred years ago, before the carbon dioxide emissions of the industrial revolution, nearly all coral reefs are found in the red-colored regions with a few in the orange and and very few in the yellow-green regions. No corals are found in the more blue and purple colored regions.
Year 1994: Already, as a result of historical carbon dioxide emissions, the area that is most suitable for coral growth has retreated to the western Pacific Ocean (and a little bit of the Indian Ocean). Most existing corals are already in marginal environments for coral growth.
Year 2040: Already, there is no place left in the ocean that is optimal for coral growth. In parts of the Southern Ocean, shells of some organisms, such as pteropods, are starting to dissolve.
Year 2099: By the end of the century, there is no place left in the ocean with the kind of ocean chemistry where corals are found growing naturally. Shells of marine organisms are dissolving through most of the Southern Ocean. Credit: Ken Caldeira, Testimony to the House Natural Resources Committee, Subcommittee on Fisheries, Wildlife and Oceans: Hearing on "Wildlife and Oceans in a Changing Climate."
References:
Cao, L., and K. Caldeira (2008), "Atmospheric CO2 stabilization and ocean acidification", Geophys. Res. Lett., doi:10.1029/2008GL035072, in press.
House Natural Resources Committee, Subcommittee on Fisheries, Wildlife and Oceans: Hearing on "Wildlife and Oceans in a Changing Climate." Written testimony by Dr. Ken Caldeira, Ph.D., Department of Global Ecology, Carnegie Institution of Washington: "Climate Change and Acidification are Affecting our Oceans" [*.pdf] - April 17, 2007.
Ocean acidification could devastate coral reefs and other marine ecosystems even if atmospheric carbon dioxide stabilizes at 450 ppm, a level well below that of many climate change forecasts, report chemical oceanographers Long Cao and Ken Caldeira of the Carnegie Institution's Department of Global Ecology in the journal Geophysical Research Letters.
The researchers' conclusions are based on computer simulations of ocean chemistry stabilized at atmospheric CO2 levels ranging from 280 parts per million (pre-industrial levels) to 2000 ppm. Present levels are 380 ppm and rapidly rising due to accelerating emissions from human activities, primarily the burning of fossil fuels.
This study was initiated as a result of Caldeira's testimony [*.pdf] before a Congressional subcommittee on Fisheries, Wildlife and Oceans in April of 2007. At that time he was asked what stabilization level would be needed to preserve the marine environment, but had to answer that no such study had yet addressed that question. Cao and Caldeira's study helps fill the gap.
Atmospheric CO2 absorbed by the oceans' surface water produces carbonic acid, the same acid that gives soft drinks their fizz, making certain carbonate minerals dissolve more readily in seawater. This is especially true for aragonite, the mineral used by corals and many other marine organisms to grow their skeletons. For corals to be able to build reefs, which requires rapid growth and strong skeletons, the surrounding water needs to be highly supersaturated with aragonite.
Before the industrial revolution, over 98% of warm water coral reefs were surrounded by open ocean waters at least 3.5 times supersaturated with aragonite. But even if atmospheric CO2 stabilizes at the current level of 380 ppm, fewer than half of existing coral reef will remain in such an environment. If the levels stabilize at 450 ppm, fewer than 10% of reefs would be in waters with the kind of chemistry that has sustained coral reefs in the past. - Long CaoFor the ecologically productive cold waters near the poles, the prospects are equally grim, says Cao. At atmospheric CO2 levels as low as 450 ppm, large parts of the Southern Ocean, the Arctic Ocean, and the North Pacific would experience a rise in acidity that would violate US Environmental Protection Agency water quality standards. Under those conditions the shells of many marine organisms would dissolve, including those at the base of the food chain:
 energy :: sustainability :: biomass :: bioenergy :: fossil fuels :: carbon dioxide :: ocean acidification :: marine biodiversity :: oceans ::
energy :: sustainability :: biomass :: bioenergy :: fossil fuels :: carbon dioxide :: ocean acidification :: marine biodiversity :: oceans :: If current trends in CO2 emissions continue unabated, says Caldeira, in the next few decades, we will produce chemical conditions in the oceans that have not been seen for tens of millions of years. We are doing something very profound to our oceans. Ecosystems like coral reefs that have been around for many millions of years just won't be able to cope with the change.
When you go to the seashore, the oceans seem huge. It's hard to imagine we could wreck it all. But if we want our children to enjoy a healthy ocean, we need to start cutting carbon emissions now. - Ken CaldeiraThe Carnegie Institution has been a pioneering force in basic scientific research since 1902. It is a private, nonprofit organization with six research departments throughout the U.S. Carnegie scientists are leaders in plant biology, developmental biology, astronomy, materials science, global ecology, and Earth and planetary science. The Department of Global Ecology, located in Stanford, California, was established in 2002 to help build the scientific foundations for a sustainable future. Its scientists conduct basic research on a wide range of large-scale environmental issues, including climate change, ocean acidification, biological invasions, and changes in biodiversity.
Figure: Maps showing the distribution of ocean chemistry suitable for coral growth for different time periods, assuming “business-as-usual” CO2 emissions. Colors represent the chemical force promoting the development of coral skeletons.
Year 1765: Several hundred years ago, before the carbon dioxide emissions of the industrial revolution, nearly all coral reefs are found in the red-colored regions with a few in the orange and and very few in the yellow-green regions. No corals are found in the more blue and purple colored regions.
Year 1994: Already, as a result of historical carbon dioxide emissions, the area that is most suitable for coral growth has retreated to the western Pacific Ocean (and a little bit of the Indian Ocean). Most existing corals are already in marginal environments for coral growth.
Year 2040: Already, there is no place left in the ocean that is optimal for coral growth. In parts of the Southern Ocean, shells of some organisms, such as pteropods, are starting to dissolve.
Year 2099: By the end of the century, there is no place left in the ocean with the kind of ocean chemistry where corals are found growing naturally. Shells of marine organisms are dissolving through most of the Southern Ocean. Credit: Ken Caldeira, Testimony to the House Natural Resources Committee, Subcommittee on Fisheries, Wildlife and Oceans: Hearing on "Wildlife and Oceans in a Changing Climate."
References:
Cao, L., and K. Caldeira (2008), "Atmospheric CO2 stabilization and ocean acidification", Geophys. Res. Lett., doi:10.1029/2008GL035072, in press.
House Natural Resources Committee, Subcommittee on Fisheries, Wildlife and Oceans: Hearing on "Wildlife and Oceans in a Changing Climate." Written testimony by Dr. Ken Caldeira, Ph.D., Department of Global Ecology, Carnegie Institution of Washington: "Climate Change and Acidification are Affecting our Oceans" [*.pdf] - April 17, 2007.
 --------------
--------------
 Mongabay, a leading resource for news and perspectives on environmental and conservation issues related to the tropics, has launched Tropical Conservation Science - a new, open access academic e-journal. It will cover a wide variety of scientific and social studies on tropical ecosystems, their biodiversity and the threats posed to them.
Mongabay, a leading resource for news and perspectives on environmental and conservation issues related to the tropics, has launched Tropical Conservation Science - a new, open access academic e-journal. It will cover a wide variety of scientific and social studies on tropical ecosystems, their biodiversity and the threats posed to them.









0 Comments:
Post a Comment
Links to this post:
Create a Link
<< Home