
 Austrian bioenergy group Cycleenergy acquired controlling interest in Greenpower Projektentwicklungs GmbH, expanding its biomass operational portfolio by 16 MW to a total of 22 MW. In the transaction Cycleenergy took over 51% of the company and thereby formed a joint venture with Porr Infrastruktur GmbH, a subsidiary of Austrian construction company Porr AG. Greenpower operates two wood chip CHP facilities in Upper and Lower Austria, each with an electric capacity of 2 MW. The plants have been in operation since the middle of last year and consume more than 30,000 tonnes of wood chips and are expected to generate over €5 million in additional revenue.
Cycleenergy - February 6, 2007. Austrian bioenergy group Cycleenergy acquired controlling interest in Greenpower Projektentwicklungs GmbH, expanding its biomass operational portfolio by 16 MW to a total of 22 MW. In the transaction Cycleenergy took over 51% of the company and thereby formed a joint venture with Porr Infrastruktur GmbH, a subsidiary of Austrian construction company Porr AG. Greenpower operates two wood chip CHP facilities in Upper and Lower Austria, each with an electric capacity of 2 MW. The plants have been in operation since the middle of last year and consume more than 30,000 tonnes of wood chips and are expected to generate over €5 million in additional revenue.
Cycleenergy - February 6, 2007.
 The 2008 edition of Bioenergy World Europe will take place in Verona, Italy, from 7 to 10 February. Gathering a broad range of international exhibitors covering gaseous, liquid and solid bioenergy, the event aims to offer participants the possibility of developing their business through meetings with professionals, thematic study tours and an international forum focusing on market and regulatory issues, as well as industry expertise.
Bioenergy World Europe - February 5, 2007. The 2008 edition of Bioenergy World Europe will take place in Verona, Italy, from 7 to 10 February. Gathering a broad range of international exhibitors covering gaseous, liquid and solid bioenergy, the event aims to offer participants the possibility of developing their business through meetings with professionals, thematic study tours and an international forum focusing on market and regulatory issues, as well as industry expertise.
Bioenergy World Europe - February 5, 2007.
 The World GTL Summit will take place between 12 – 14th May 2008 in London. Key topics to be discussed include: the true value of Gas-to-Liquids (GTL) projects, well-to-wheels analyses of the GTL value chain; construction, logistics and procurement challenges; the future for small-scale Fischer-Tropsch (FT) projects; Technology, economics, politics and logistics of Coal-to-Liquids (CTL); latest Biomass-to-Liquids (BTL) commercialisation initiatives.
CWC Exhibitions - February 4, 2007. The World GTL Summit will take place between 12 – 14th May 2008 in London. Key topics to be discussed include: the true value of Gas-to-Liquids (GTL) projects, well-to-wheels analyses of the GTL value chain; construction, logistics and procurement challenges; the future for small-scale Fischer-Tropsch (FT) projects; Technology, economics, politics and logistics of Coal-to-Liquids (CTL); latest Biomass-to-Liquids (BTL) commercialisation initiatives.
CWC Exhibitions - February 4, 2007.
 The 4th Annual Brussels Climate Change Conference is announced for 26 - 27 February 2008. This joint CEPS/Epsilon conference will explore the key issues for a post-Kyoto agreement on climate change. The conference focuses on EU and global issues relating to global warming, and in particular looks at the following issues: - Post-2012 after Bali and before the Hokkaido G8 summit; Progress of EU integrated energy and climate package, burden-sharing renewables and technology; EU Emissions Trading Review with a focus on investment; Transport
Climatepolicy.eu - January 28, 2007. The 4th Annual Brussels Climate Change Conference is announced for 26 - 27 February 2008. This joint CEPS/Epsilon conference will explore the key issues for a post-Kyoto agreement on climate change. The conference focuses on EU and global issues relating to global warming, and in particular looks at the following issues: - Post-2012 after Bali and before the Hokkaido G8 summit; Progress of EU integrated energy and climate package, burden-sharing renewables and technology; EU Emissions Trading Review with a focus on investment; Transport
Climatepolicy.eu - January 28, 2007.
 Japan's Marubeni Corp. plans to begin importing a bioethanol compound from Brazil for use in biogasoline sold by petroleum wholesalers in Japan. The trading firm will import ETBE, which is synthesized from petroleum products and ethanol derived from sugar cane. The compound will be purchased from Brazilian petrochemical company Companhia Petroquimica do Sul and in February, Marubeni will supply 6,500 kilolitres of the ETBE, worth around US$7 million, to a biogasoline group made up of petroleum wholesalers. Wholesalers have been introducing biofuels since last April by mixing 7 per cent ETBE into gasoline. Plans call for 840 million liters of ETBE to be procured annually from domestic and foreign suppliers by 2010.
Trading Markets - January 24, 2007. Japan's Marubeni Corp. plans to begin importing a bioethanol compound from Brazil for use in biogasoline sold by petroleum wholesalers in Japan. The trading firm will import ETBE, which is synthesized from petroleum products and ethanol derived from sugar cane. The compound will be purchased from Brazilian petrochemical company Companhia Petroquimica do Sul and in February, Marubeni will supply 6,500 kilolitres of the ETBE, worth around US$7 million, to a biogasoline group made up of petroleum wholesalers. Wholesalers have been introducing biofuels since last April by mixing 7 per cent ETBE into gasoline. Plans call for 840 million liters of ETBE to be procured annually from domestic and foreign suppliers by 2010.
Trading Markets - January 24, 2007.
 Toyota Tsusho Corp., Ohta Oil Mill Co. and Toyota Chemical Engineering Co., say it and two other firms have jointly developed a technology to produce biodiesel fuel at lower cost. Biodiesel is made by blending methanol into plant-derived oil. The new technology requires smaller amounts of methanol and alkali catalysts than conventional technologies. In addition, the new technology makes water removal facilities unnecessary.
JCN Network - January 22, 2007. Toyota Tsusho Corp., Ohta Oil Mill Co. and Toyota Chemical Engineering Co., say it and two other firms have jointly developed a technology to produce biodiesel fuel at lower cost. Biodiesel is made by blending methanol into plant-derived oil. The new technology requires smaller amounts of methanol and alkali catalysts than conventional technologies. In addition, the new technology makes water removal facilities unnecessary.
JCN Network - January 22, 2007.
 Finland's Metso Paper and SWISS COMBI - W. Kunz dryTec A.G. have entered a licence agreement for the SWISS COMBI belt dryer KUVO, which allows biomass to be dried in a low temperature environment and at high capacity, both for pulp & paper and bioenergy applications.
Kauppalehti - January 22, 2007. Finland's Metso Paper and SWISS COMBI - W. Kunz dryTec A.G. have entered a licence agreement for the SWISS COMBI belt dryer KUVO, which allows biomass to be dried in a low temperature environment and at high capacity, both for pulp & paper and bioenergy applications.
Kauppalehti - January 22, 2007.
 Record warm summers cause extreme ice melt in Greenland: an international team of scientists, led by Dr Edward Hanna at the University of Sheffield, has found that recent warm summers have caused the most extreme Greenland ice melting in 50 years. The new research provides further evidence of a key impact of global warming and helps scientists place recent satellite observations of Greenland´s shrinking ice mass in a longer-term climatic context. Findings are published in the 15 January 2008 issue of Journal of Climate.
University of Sheffield - January 15, 2007. Record warm summers cause extreme ice melt in Greenland: an international team of scientists, led by Dr Edward Hanna at the University of Sheffield, has found that recent warm summers have caused the most extreme Greenland ice melting in 50 years. The new research provides further evidence of a key impact of global warming and helps scientists place recent satellite observations of Greenland´s shrinking ice mass in a longer-term climatic context. Findings are published in the 15 January 2008 issue of Journal of Climate.
University of Sheffield - January 15, 2007.
 Japan's Tsukishima Kikai Co. and Marubeni Corp. have together clinched an order from Oenon Holdings Inc. for a plant that will make bioethanol from rice. The Oenon group will invest around 4.4 billion yen (US$40.17 million) in the project, half of which will be covered by a subsidy from the Ministry of Agriculture, Forestry and Fisheries. The plant will initially produce bioethanol from imported rice, with plans to use Hokkaido-grown rice in the future. It will produce 5 million liters per year starting in 2009, increasing output to 15m liters in 2011. The facility will be able to produce as much as 50,000 liters of bioethanol from 125 tons of rice each day.
Trading Markets - January 11, 2007. Japan's Tsukishima Kikai Co. and Marubeni Corp. have together clinched an order from Oenon Holdings Inc. for a plant that will make bioethanol from rice. The Oenon group will invest around 4.4 billion yen (US$40.17 million) in the project, half of which will be covered by a subsidy from the Ministry of Agriculture, Forestry and Fisheries. The plant will initially produce bioethanol from imported rice, with plans to use Hokkaido-grown rice in the future. It will produce 5 million liters per year starting in 2009, increasing output to 15m liters in 2011. The facility will be able to produce as much as 50,000 liters of bioethanol from 125 tons of rice each day.
Trading Markets - January 11, 2007.
 PetroSun, Inc. announced today that its subsidiary, PetroSun BioFuels Refining, has entered into a JV to construct and operate a biodiesel refinery near Coolidge, Arizona. The feedstock for the refinery will be algal oil produced by PetroSun BioFuels at algae farms to be located in Arizona. The refinery will have a capacity of thirty million gallons and will produce 100% renewable biodiesel. PetroSun BioFuels will process the residual algae biomass into ethanol.
MarketWire - January 10, 2007. PetroSun, Inc. announced today that its subsidiary, PetroSun BioFuels Refining, has entered into a JV to construct and operate a biodiesel refinery near Coolidge, Arizona. The feedstock for the refinery will be algal oil produced by PetroSun BioFuels at algae farms to be located in Arizona. The refinery will have a capacity of thirty million gallons and will produce 100% renewable biodiesel. PetroSun BioFuels will process the residual algae biomass into ethanol.
MarketWire - January 10, 2007.
 BlueFire Ethanol Fuels Inc, which develops and operates carbohydrate-based transportation fuel production facilities, has secured capital liquidity for corporate overhead and continued project development in the value of US$15 million with Quercus, an environmentally focused trust.
BlueFire Ethanol Fuels - January 09, 2007. BlueFire Ethanol Fuels Inc, which develops and operates carbohydrate-based transportation fuel production facilities, has secured capital liquidity for corporate overhead and continued project development in the value of US$15 million with Quercus, an environmentally focused trust.
BlueFire Ethanol Fuels - January 09, 2007.
 Some $170 billion in new technology development projects, infrastructure equipment and construction, and biofuel refineries will result from the ethanol production standards contained the new U.S. Energy Bill, says BIO, the global Biotechnology Industry Organization. According to Brent Erickson, BIO's executive vice president "Such a new energy infrastructure has not occurred in more than 100 years. We are at the point where we were in the 1850s when kerosene was first distilled and began to replace whale oil. This technology will be coming so fast that what we say today won't be true in two years."
Chemical & Engineering News - January 07, 2007. Some $170 billion in new technology development projects, infrastructure equipment and construction, and biofuel refineries will result from the ethanol production standards contained the new U.S. Energy Bill, says BIO, the global Biotechnology Industry Organization. According to Brent Erickson, BIO's executive vice president "Such a new energy infrastructure has not occurred in more than 100 years. We are at the point where we were in the 1850s when kerosene was first distilled and began to replace whale oil. This technology will be coming so fast that what we say today won't be true in two years."
Chemical & Engineering News - January 07, 2007.
 Scottish and Southern Energy plc, the UK's second largest power company, has completed the acquisition of Slough Heat and Power Ltd from SEGRO plc for a total cash consideration of £49.25m. The 101MW CHP plant is the UK’s largest dedicated biomass energy facility fueled by wood chips, biomass and waste paper. Part of the plant is contracted under the Non Fossil Fuel Obligation and part of it produces over 200GWH of output qualifying for Renewable Obligation Certificates (ROCs), which is equivalent to around 90MW of wind generation.
Scottish & Southern Energy - January 2, 2007. Scottish and Southern Energy plc, the UK's second largest power company, has completed the acquisition of Slough Heat and Power Ltd from SEGRO plc for a total cash consideration of £49.25m. The 101MW CHP plant is the UK’s largest dedicated biomass energy facility fueled by wood chips, biomass and waste paper. Part of the plant is contracted under the Non Fossil Fuel Obligation and part of it produces over 200GWH of output qualifying for Renewable Obligation Certificates (ROCs), which is equivalent to around 90MW of wind generation.
Scottish & Southern Energy - January 2, 2007.
 PetroChina Co Ltd, the country's largest oil and gas producer, plans to invest 800 million yuan to build an ethanol plant in Nanchong, in the southwestern province of Sichuan, its parent China National Petroleum Corp said. The ethanol plant has a designed annual capacity of 100,000 tons.
ABCMoneyNews - December 21, 2007. PetroChina Co Ltd, the country's largest oil and gas producer, plans to invest 800 million yuan to build an ethanol plant in Nanchong, in the southwestern province of Sichuan, its parent China National Petroleum Corp said. The ethanol plant has a designed annual capacity of 100,000 tons.
ABCMoneyNews - December 21, 2007.
 Mexico passed legislation to promote biofuels last week, offering unspecified support to farmers that grow crops for the production of any renewable fuel. Agriculture Minister Alberto Cardenas said Mexico could expand biodiesel faster than ethanol. More soon.
Reuters - December 20, 2007. Mexico passed legislation to promote biofuels last week, offering unspecified support to farmers that grow crops for the production of any renewable fuel. Agriculture Minister Alberto Cardenas said Mexico could expand biodiesel faster than ethanol. More soon.
Reuters - December 20, 2007.
 Oxford Catalysts has placed an order worth approximately €700,000 (US$1 million) with the German company Amtec for the purchase of two Spider16 high throughput screening reactors. The first will be used to speed up the development of catalysts for hydrodesulphurisation (HDS). The second will be used to further the development of catalysts for use in gas to liquid (GTL) and Fischer-Tropsch processes which can be applied to next generation biofuels.
AlphaGalileo - December 18, 2007. Oxford Catalysts has placed an order worth approximately €700,000 (US$1 million) with the German company Amtec for the purchase of two Spider16 high throughput screening reactors. The first will be used to speed up the development of catalysts for hydrodesulphurisation (HDS). The second will be used to further the development of catalysts for use in gas to liquid (GTL) and Fischer-Tropsch processes which can be applied to next generation biofuels.
AlphaGalileo - December 18, 2007.
 According to the Instituto Brasileiro de Geografia e Estatística (IBGE), Brazil's production of sugarcane will increase from 514,1 million tonnes this season, to a record 561,8 million tonnes in the 2008/09 cyclus - an increase of 9.3%. New numbers are also out for the 2007 harvest in Brazil's main sugarcane growing region, the Central-South: a record 425 million tonnes compared to 372,7 million tonnes in 2006, or a 14% increase. The estimate was provided by Unica – the União da Indústria de Cana-de-Açúcar.
Jornal Cana - December 16, 2007. According to the Instituto Brasileiro de Geografia e Estatística (IBGE), Brazil's production of sugarcane will increase from 514,1 million tonnes this season, to a record 561,8 million tonnes in the 2008/09 cyclus - an increase of 9.3%. New numbers are also out for the 2007 harvest in Brazil's main sugarcane growing region, the Central-South: a record 425 million tonnes compared to 372,7 million tonnes in 2006, or a 14% increase. The estimate was provided by Unica – the União da Indústria de Cana-de-Açúcar.
Jornal Cana - December 16, 2007.
 The University of East Anglia and the UK Met Office's Hadley Centre have today released preliminary global temperature figures for 2007, which show the top 11 warmest years all occurring in the last 13 years. The provisional global figure for 2007 using data from January to November, currently places the year as the seventh warmest on records dating back to 1850. The announcement comes as the Secretary-General of the World Meteorological Organization (WMO), Michel Jarraud, speaks at the Conference of the Parties (COP) in Bali.
Eurekalert - December 13, 2007. The University of East Anglia and the UK Met Office's Hadley Centre have today released preliminary global temperature figures for 2007, which show the top 11 warmest years all occurring in the last 13 years. The provisional global figure for 2007 using data from January to November, currently places the year as the seventh warmest on records dating back to 1850. The announcement comes as the Secretary-General of the World Meteorological Organization (WMO), Michel Jarraud, speaks at the Conference of the Parties (COP) in Bali.
Eurekalert - December 13, 2007.
 The Royal Society of Chemistry has announced it will launch a new journal in summer 2008, Energy & Environmental Science, which will distinctly address both energy and environmental issues. In recognition of the importance of research in this subject, and the need for knowledge transfer between scientists throughout the world, from launch the RSC will make issues of Energy & Environmental Science available free of charge to readers via its website, for the first 18 months of publication. This journal will highlight the important role that the chemical sciences have in solving the energy problems we are facing today. It will link all aspects of energy and the environment by publishing research relating to energy conversion and storage, alternative fuel technologies, and environmental science.
AlphaGalileo - December 10, 2007. The Royal Society of Chemistry has announced it will launch a new journal in summer 2008, Energy & Environmental Science, which will distinctly address both energy and environmental issues. In recognition of the importance of research in this subject, and the need for knowledge transfer between scientists throughout the world, from launch the RSC will make issues of Energy & Environmental Science available free of charge to readers via its website, for the first 18 months of publication. This journal will highlight the important role that the chemical sciences have in solving the energy problems we are facing today. It will link all aspects of energy and the environment by publishing research relating to energy conversion and storage, alternative fuel technologies, and environmental science.
AlphaGalileo - December 10, 2007.
 Dutch researcher Bas Bougie has developed a laser system to investigate soot development in diesel engines. Small soot particles are not retained by a soot filter but are, however, more harmful than larger soot particles. Therefore, soot development needs to be tackled at the source. Laser Induced Incandescence is a technique that reveals exactly where soot is generated and can be used by project partners to develop cleaner diesel engines. Terry Meyer, an Iowa State University assistant professor of mechanical engineering, is using similar laser technology to develop advanced sensors capable of screening the combustion behavior and soot characteristics specifically of biofuels.
Eurekalert - December 7, 2007. Dutch researcher Bas Bougie has developed a laser system to investigate soot development in diesel engines. Small soot particles are not retained by a soot filter but are, however, more harmful than larger soot particles. Therefore, soot development needs to be tackled at the source. Laser Induced Incandescence is a technique that reveals exactly where soot is generated and can be used by project partners to develop cleaner diesel engines. Terry Meyer, an Iowa State University assistant professor of mechanical engineering, is using similar laser technology to develop advanced sensors capable of screening the combustion behavior and soot characteristics specifically of biofuels.
Eurekalert - December 7, 2007.
 Lithuania's first dedicated biofuel terminal has started operating in Klaipeda port. At the end of November 2007, the stevedoring company Vakaru krova (VK) started activities to manage transshipments. The infrastructure of the biodiesel complex allows for storage of up to 4000 cubic meters of products. During the first year, the terminal plans to transship about 70.000 tonnes of methyl ether, after that the capacities of the terminal would be increased. Investments to the project totaled €2.3 million.
Agrimarket - December 5, 2007. Lithuania's first dedicated biofuel terminal has started operating in Klaipeda port. At the end of November 2007, the stevedoring company Vakaru krova (VK) started activities to manage transshipments. The infrastructure of the biodiesel complex allows for storage of up to 4000 cubic meters of products. During the first year, the terminal plans to transship about 70.000 tonnes of methyl ether, after that the capacities of the terminal would be increased. Investments to the project totaled €2.3 million.
Agrimarket - December 5, 2007.
 New Holland supports the use of B100 biodiesel in all equipment with New Holland-manufactured diesel engines, including electronic injection engines with common rail technology. Overall, nearly 80 percent of the tractor and equipment manufacturer's New Holland-branded products with diesel engines are now available to operate on B100 biodiesel. Tractor and equipment maker John Deere meanwhile clarified its position for customers that want to use biodiesel blends up to B20.
Grainnet - December 5, 2007. New Holland supports the use of B100 biodiesel in all equipment with New Holland-manufactured diesel engines, including electronic injection engines with common rail technology. Overall, nearly 80 percent of the tractor and equipment manufacturer's New Holland-branded products with diesel engines are now available to operate on B100 biodiesel. Tractor and equipment maker John Deere meanwhile clarified its position for customers that want to use biodiesel blends up to B20.
Grainnet - December 5, 2007.
 According to Wetlands International, an NGO, the Kyoto Protocol as it currently stands does not take into account possible emissions from palm oil grown on a particular type of land found in Indonesia and Malaysia, namely peatlands.
Mongabay - December 5, 2007. According to Wetlands International, an NGO, the Kyoto Protocol as it currently stands does not take into account possible emissions from palm oil grown on a particular type of land found in Indonesia and Malaysia, namely peatlands.
Mongabay - December 5, 2007.
 Malaysia's oil & gas giant Petronas considers entering the biofuels sector. Zamri Jusoh, senior manager of Petronas' petroleum development management unit told reporters "of course our focus is on oil and gas, but I think as we move into the future we cannot ignore the importance of biofuels."
AFP - December 5, 2007. Malaysia's oil & gas giant Petronas considers entering the biofuels sector. Zamri Jusoh, senior manager of Petronas' petroleum development management unit told reporters "of course our focus is on oil and gas, but I think as we move into the future we cannot ignore the importance of biofuels."
AFP - December 5, 2007.

|
|
|
|
|
 A type of bacteria that helps termites digest wood could be key to making ethanol cheaply from non-food crops such as wood and grass. ZeaChem, a startup based in Menlo Park, California, has developed a hybrid biochemical and thermochemical process that utilizes all fractions of the biomass feed and converts it with Moorella thermoacetica. The process can yield 50 percent more ethanol from a given amount of biomass than conventional processes. The net energy ratio of biofuel produced this way is between 10 and 12, compared with first generation biofuels like corn ethanol, which come in at around 1.5. The new net energy ratio benchmark radically changes any biofuel policy debate.
A type of bacteria that helps termites digest wood could be key to making ethanol cheaply from non-food crops such as wood and grass. ZeaChem, a startup based in Menlo Park, California, has developed a hybrid biochemical and thermochemical process that utilizes all fractions of the biomass feed and converts it with Moorella thermoacetica. The process can yield 50 percent more ethanol from a given amount of biomass than conventional processes. The net energy ratio of biofuel produced this way is between 10 and 12, compared with first generation biofuels like corn ethanol, which come in at around 1.5. The new net energy ratio benchmark radically changes any biofuel policy debate.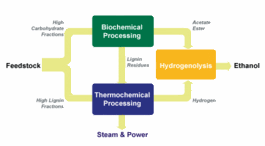 ZeaChem's approach to biorefining uses a combination of biochemical and thermochemical processing steps (schematic, click to enlarge). The hybrid process improves yield by making more efficient use of biomass than conventional techniques do. It begins, as do other techniques for making ethanol, with breaking down biomass into sugars. At this point, conventional processes use yeast to ferment the sugars into ethanol. But this process is wasteful: about a third of the carbon in the sugars never makes it into the fuel. Instead, it's released into the atmosphere as carbon dioxide.
ZeaChem's approach to biorefining uses a combination of biochemical and thermochemical processing steps (schematic, click to enlarge). The hybrid process improves yield by making more efficient use of biomass than conventional techniques do. It begins, as do other techniques for making ethanol, with breaking down biomass into sugars. At this point, conventional processes use yeast to ferment the sugars into ethanol. But this process is wasteful: about a third of the carbon in the sugars never makes it into the fuel. Instead, it's released into the atmosphere as carbon dioxide. energy :: sustainability :: biomass :: bioenergy :: biofuels :: ethanol :: gasification :: fermentation :: lignocellulose :: efficiency ::
energy :: sustainability :: biomass :: bioenergy :: biofuels :: ethanol :: gasification :: fermentation :: lignocellulose :: efficiency ::  --------------
--------------
 Austrian bioenergy group Cycleenergy acquired controlling interest in Greenpower Projektentwicklungs GmbH, expanding its biomass operational portfolio by 16 MW to a total of 22 MW. In the transaction Cycleenergy took over 51% of the company and thereby formed a joint venture with Porr Infrastruktur GmbH, a subsidiary of Austrian construction company Porr AG. Greenpower operates two wood chip CHP facilities in Upper and Lower Austria, each with an electric capacity of 2 MW. The plants have been in operation since the middle of last year and consume more than 30,000 tonnes of wood chips and are expected to generate over €5 million in additional revenue.
Austrian bioenergy group Cycleenergy acquired controlling interest in Greenpower Projektentwicklungs GmbH, expanding its biomass operational portfolio by 16 MW to a total of 22 MW. In the transaction Cycleenergy took over 51% of the company and thereby formed a joint venture with Porr Infrastruktur GmbH, a subsidiary of Austrian construction company Porr AG. Greenpower operates two wood chip CHP facilities in Upper and Lower Austria, each with an electric capacity of 2 MW. The plants have been in operation since the middle of last year and consume more than 30,000 tonnes of wood chips and are expected to generate over €5 million in additional revenue.








0 Comments:
Post a Comment
Links to this post:
Create a Link
<< Home